Busting common myths about hepatitis C treatment in Canada

Highly effective medications that can cure hepatitis C infection, called direct-acting antivirals (DAAs), have been approved for use in Canada for people over the age of 18 since 2014. These medications are well-tolerated, nearly 100% effective, and have treatment durations of just eight or 12 weeks. DAAs have simplified hepatitis C treatment considerably, allowing treatments to be prescribed by primary care providers, such as nurse practitioners and family doctors.
Now that the treatment itself is so simple, getting coverage for reimbursement of DAAs is the most complicated and challenging aspect of treating hepatitis C infection in Canada. Almost 90% of DAA claims are covered by public drug plans in Canada, and because publicly funded prescription drug coverage in Canada is so complicated, there are many misunderstandings about how DAAs are covered, both among patients and providers. In fact, medication coverage for DAAs is so complicated, a key role of patient support programs created by manufacturers is helping patients figure out and apply for reimbursement approval. This has resulted in misconceptions about DAAs, many of which contribute to barriers to accessing care among people affected by hepatitis C, particularly for Indigenous people and people who use drugs.
Through my research, as well as my work with healthcare policy-makers and providers, I have encountered many of these misconceptions and myths, and I have summarized the four most common below, outlining the facts and evidence that dispel them. Ensuring everyone has a correct understanding of how DAAs are covered by public drug plans in Canada is one way we can try to break down barriers to care and reduce health inequities for people affected by hepatitis C.
Myth 1: When a hepatitis C drug is approved by Health Canada, everyone in Canada can access it
There is often a lot of media coverage and attention whenever a new drug product is approved by Health Canada. This approval only means that a drug product is allowed to be marketed and sold in Canada. It takes an average of one year from when a new drug is approved by Health Canada to when it is included on lists of medications covered by public drug plans, also known as public formularies. This has been found to be due to the additional reviews that are required before public drug plans decide whether to cover a new medicine.
Before a publicly funded drug plan can consider adding a drug to their formulary that Health Canada has newly approved, the drug must go through an assessment for clinical and cost-effectiveness. Known as a reimbursement review, these are conducted by the Canadian Agency for Drugs and Technologies in Health (CADTH) , or in Quebec by the Institut national d’excellence en santé et en services sociaux (INESSS). Once a reimbursement review has been conducted, if a product is not found to be cost-effective, a negotiation process is undertaken through the pan-Canadian Pharmaceutical Alliance (pCPA) to attempt to reduce the cost. If an agreement is reached that allows the product to be deemed cost-effective, manufacturers must then submit a separate request to each of the 13 provincial and territorial and six federal publicly funded drug plans to consider their product to be listed as a benefit.
Additionally, there are variations in the criteria for reimbursement applied by each publicly funded drug plan when they eventually add a product to their formulary, including different clinical and administrative criteria. The length of time from when a drug is initially approved by Health Canada to when it is first listed as a Public Drug Benefit by each province can also vary widely. For sofosbuvir/velpatasvir (Epclusa) the shortest time was 7 months (listed in Ontario) and the longest was 9 months (listed in Nova Scotia), as seen in figure 1. For glecaprevir/pibrentasvir (Maviret), the shortest time was 17 months (listed in British Columbia) and the longest was 28 months (listed in Newfoundland and Labrador).
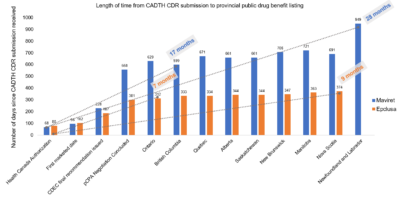
| Number of days since submission received | ||
|---|---|---|
| Milestones in progress to become available in Canada | Maviret | Epclusa |
| CADTH CDR submission received (pre NOC) | 0 | 0 |
| Health Canada Authorization | 68 | 80 |
| First marketed date | 96 | 102 |
| CDEC final recommendation issued | 228 | 187 |
| pCPA Negotiation Concluded | 558 | 301 |
| Ontario | 629 | 312 |
| British Columbia | 599 | 333 |
| Quebec | 671 | 334 |
| Alberta | 661 | 344 |
| Saskatchewan | 661 | 344 |
| New Brunswick | 706 | 347 |
| Manitoba | 721 | 363 |
| Nova Scotia | 691 | 374 |
| Newfoundland and Labrador | 949 | |
Myth 2: Hepatitis C treatment with DAAs costs $50,000 per treatment course
The pCPA facilitates negotiation of the list price for brand-name drug products in Canada jointly among all 13 provincial and territorial governments, the federal government and drug manufacturers. However, governments across Canada also undertake their own individual negotiations with manufacturers when they agree to list a product on their plan, which are often utilized to further reduce the final costs. The agreements reached in these negotiations, known as product listing agreements), are strictly confidential and governments are bound by non-disclosure agreements.
Often product listing agreements stipulate that the public drug plan will pay the list price for a drug product but will then be reimbursed by the manufacturer for an agreed upon amount, depending on the fulfilment of certain criteria. These rebate contracts effectively reduce the final cost of a drug product for publicly funded drug plans, however there is limited transparency around this final cost, which is almost certainly markedly lower than the list price.
The opacity and secrecy of product listing agreements, particularly for drug products that are publicized as ‘high-cost drugs’, result in confusion among the general public, as well as prescribers themselves. They are only ever aware of the listed price of a drug product, not the actual cost to the public drug plan once any agreed-upon rebates are paid by the manufacturer. For example, the average cost paid by Canadian public drug plans per patient (i.e. the list price) for glecaprevir/pibrentasvir (Maviret) tablets was reported to be $41,088 in 2019. However, this is not necessarily the final cost that public drug plans may end up paying per patient treated with glecaprevir/pibrentasvir (Maviret), and often cost savings become larger as the number of patients treated increases.
Myth 3: DAA treatment is available in Canada for all children aged three to 12 years
A new granule formula of glecaprevir/pibrentasvir (Maviret) received authorization by Health Canada for treatment of hepatitis C infection in children aged three to 12 years on April 7, 2022. While CADTH recently published a rapid review of DAAs for pediatric chronic hepatitis C virus infection, there was no reimbursement review listed by CADTH or by INESSS in progress for glecaprevir/pibrentasvir (Maviret) granules as of February 24, 2024. A request for reimbursement review must be made directly by a manufacturer if they want publicly funded drug plans to cover their drug, and they are required to pay a sponsorship fee to both CADTH and INESSS. Public drug plans may choose independently to review a drug that has not yet had a CADTH or INESSS reimbursement review completed. However, the manufacturer of a drug product that has not undergone CADTH or INESSS reimbursement review is unable to request for any public drugs plan in Canada to consider their product for listing as a benefit.
Since this new formulation was approved by Health Canada, pediatric patients in Canada with chronic hepatitis C infection have only been able to receive DAA treatment through exceptional coverage, clinical trials, or by paying the full cost of the medication. Those patients who are unable to afford the full cost may be able to access support through the AbbVie patient support program, also known as compassionate access. Compassionate access through manufacturers or exceptional coverage by a publicly funded drug plan often takes considerable time to be approved, and it is cumbersome and laborious for prescribers. As a result, it is likely that only a small proportion of the children aged three to 12 years in Canada with hepatitis C infection have actually been able to access DAA treatment since the granule formula of glecaprevir/pibrentasvir (Maviret) was approved by Health Canada in April 2022.
When healthcare providers share information about hepatitis C treatment for children aged three to 12 years in Canada, it is essential that they also explain that the medication is not currently listed on formularies of public drug plans, and they may require more support to find coverage. Not sharing this information may give pediatric patients affected by hepatitis C, or their families and carers, a false expectation that medications are readily available and easily accessible to them.
Myth 4: People living with chronic hepatitis C infection only have ‘one shot’ at treatment with DAAs
People living with hepatitis C infection often report that treatment providers tell them they cannot start treatment with DAAs until they have met certain benchmarks or expectations. Frequently reported benchmarks include abstinence from illicit substances, stabilization on opioid agonist therapy, or abstinence from alcohol. Providers report that these benchmarks are necessary to ensure that patients will be adherent to medications or will be able to protect themselves from reinfection from hepatitis C after being cured. While goals for treatment adherence and prevention of reinfection are important to consider when assessing treatment readiness, it’s also important that people living with hepatitis C have agency over their own healthcare. This means that their own personal goals and preferences are considered and decisions about whether DAA treatment should be commenced are made in collaboration between patients and their healthcare providers.
Providers may disagree with this and cite that it is their responsibility to ensure publicly funded medications are used appropriately. However, each drug plan determines the criteria for whether a patient is eligible for reimbursement of treatment costs, and it is the drug plans’ responsibility to determine if requests for coverage are eligible. In a study I conducted in 2023, we found that abstinence from drugs or alcohol is not listed as a criterion for DAA reimbursement eligibility by any publicly funded drug plan in Canada.
We did find that one drug plan (Newfoundland and Labrador) did not at that time cover reimbursement for subsequent DAA treatment in instances of reinfection, however six plans unequivocally do cover treatment for reinfection, and the remaining plans cover it on a case-by-case basis. Therefore, it is not accurate to say that people living with chronic hepatitis C infection only have ‘one shot’ at treatment with DAAs. The vast majority of publicly funded drug plans in Canada accept that in order to achieve hepatitis C elimination goals, it is necessary to cover DAAs for retreatment in instances of hepatitis C reinfection. Additionally, treatment of hepatitis C infection significantly reduces the risk of liver-related and all-cause mortality for individuals. While there is high awareness of the substantial cost of DAAs, there is much less awareness of the high health care costs associated with untreated chronic hepatitis C infection. So despite the cost of DAAs, treatment of both primary and subsequent hepatitis C infection is economically attractive to the public health system in Canada.
National pharmacare: The prescription to simplify drug coverage in Canada
Research on pharmacare systems in Canada has repeatedly demonstrated that our current drug coverage policies are failing across multiple metrics. Available evidence points to a universal and comprehensive public pharmacare program as the most suitable strategy to address this issue. In 2019, the Advisory Council on the Implementation of National Pharmacare handed down their final report, which recommended a national pharmacare plan that is universal, comprehensive, accessible, portable and both publicly funded and administered.
In March 2022, the Government of Canada committed to pass a Canada Pharmacare Act in 2023 and to develop a formulary and bulk purchasing plan for essential medicines by 2025. It is reported that the Liberals and New Democrats reached a deal on legislation outlining the framework to implement a universal pharmacare program, which is to be tabled in March 2024. However, there is considerable uncertainty about whether the government will actually be able to pass this act, as well as what drugs the national pharmacare plan would cover.
Any discussion about whether national pharmacare would reduce barriers to DAA access for treatment of hepatitis C infection is purely speculative, as there are not yet any studies investigating this. A single-payer system would likely reduce the time from approval by Health Canada to when all Canadians would be able to be reimbursed for the drug through their public drug plan. Additionally, given evidence from recent studies examining the impact of free medicine distribution in Ontario, there appears to be a compelling case to be made that national universal pharmacare would facilitate hepatitis C treatment access and uptake.
Sofia Bartlett, PhD, is the interim scientific director for Clinical Prevention Services at the British Columbia Centre for Disease Control. She is also an adjunct professor in the University of British Columbia School of Population and Public Health. She is an infectious disease epidemiologist and mixed methods researcher, and investigates the intersections of sexually transmitted and blood-borne infections with incarceration, substance use and digital exclusion.
